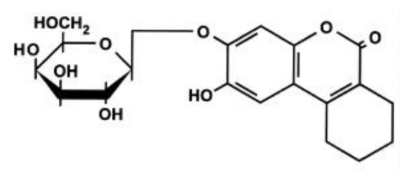
LacZ
S-Gal
S-Gal is a trademark product of Sigma Aldrich for the compound 3,4-cyclohexenoesculetin- β-D-galactopyranoside, C19H21O9Na, MW 416.35, MDL Number MFCD03458481.
S-Gal®, sodium salt is a patented water-soluble, autoclavable chromogenic substrate for β-galactosidase that is designed to replace X-Gal in blue-white selection of recombinant bacterial colonies with the lac+ phenotype. Caution: For black color development to occur, ferric ion must be present. Although we recommend ferric ammonium citrate (500 mg/L of media), other ferric compounds can be used to provide this requirement, depending upon your particular system. From Sigma-Aldrich site.
Image from Heuermann and Cosgrove PMID 11355350
Reagents ordered in February 2009
0F5879-100G, $35.54, AMMONIUM IRON(III) CITRATE, REAGENT GRADE
S7313-1G, $385.60, S-GAL(R) SODIUM SALT
S-Gal staining conditions
According to Sigma-Aldrich site:
Stock solutions of S-Gal can be made by dissolving at 50 mg/mL in deionized water, sterile-filtering and storing at -20 °C.
Bone Marrow Cells for use in MRI, From Bengtsson et al. in NHMFL reports
BMCs were harvested from C57 and ROSA26 mice and re-suspended with 1 mg/ml S-Gal (Sigma) and 0.5 mg/ml FAC (Sigma) in 1ml PBS, 0.5 mg/ml FAC in 1 ml PBS or 1 ml PBS in microcentrifuge tubes and incubated at 37°C for 2hrs. BMCs were then washed twice with PBS to remove fcaps to remove large aggregations of S-Gal and cells.
In Mouse Embryos from Kishigami et al PMID 16419090
(In brief, dissected embryos were fixed with 4% paraformaldehyde overnight, subse- quently soaked with 20% sucrose in PBS, and embedded in Optimal Cutting Temperature (O.C.T.) in liquid nitro- gen. Fourteen-lm sections were cut, washed with PBS at room temperature, soaked with 100 mM sodium phos- phate buffer (pH 7.5), 0.1% sodium deoxycholate, 0.2% Nonidet P-40, 2 mM magnesium chloride, 5 mM potas- sium ferricyanide, and 5 mM potassium ferrocyanide for 10 min at room temperature. Sections were subse- quently stained with 1 mg/ml of X-gal or S-gal at 378C. An S-gal-stained section was immediately used for in situ hybridization according to the nonradioactive protocol (Lindahl et al., 1997).
For detecting plasmids on agar plates, from Heuermann and Cosgrove PMID 11355350
For chemical analysis, ferric ammonium citrate, S-Gal, and IPTG were added to 50-mL aliquots LB broth or deionized water at final concentrations of 500, 300, and 30 µg/mL, respectively (all from Sigma). S-Gal dye was initially dissolved in N,N-dimethylformamide (DMF) (Sigma) at a concentration of 200 mg/mL and was added to water or medium for analysis at the concentration stated above before autoclaving or microwaving. For this part of the evaluation, a stock solution of S-Gal in DMF was used for convenience and for the consistent addition of S-Gal to media or aqueous solutions. S-Gal, determined empirically to be approximately 70% more soluble than X-gal at room temperature (data not shown), is suitable for the blended-medium product but requires dissolving in DMF for the preparation of more concen- trated solutions such as the stock solution described here. IPTG was added to the water or medium from an aqueous stock of 100 mg/mL. Ferric ammonium citrate was prepared as an aqueous stock solution of 200 mg/mL.
S-Gal, IPTG and ferric ammonium citrate are dry-blended with standard LB agar at the final concentrations stated above. The powdered blend was suspended in deionized water and autoclaved as previously described. For the preparation of microwaved medium, the suspended blend was heated until boiling, followed by swirling to allow the remaining agar component to go into solution. In this case, the antibiotic ampicillin was added after autoclaving (100 mg/mL).
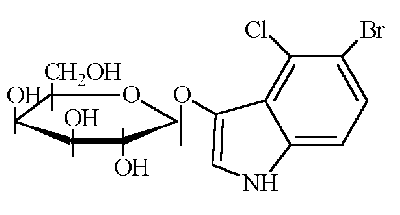
X-Gal
5-bromo-4-chloro-3-indolyl-beta-D-galactopyranoside (MW 408.63):
https://bio.davidson.edu/courses/genomics/method/xgal.gif
Cleavage by beta-galactosidase yields a dihalogen-substituted indoxyl, 5-bromo-4-chloro-3-indoxyl:
Oxidation in the presence of an electron acceptor yields a relatively insoluble indigoid dye:
Oxidants used in histochemistry
Potassium ferri- and ferrocyanide. ferricyanide is the electron acceptor; ferrocyanide is "provided in equimolar amount ... to give a system with a poised oxidation potential." (Holt and Withers, 1958, p. 524).
Nitroblue tetrazolium Used as a test for tuberculosis? PMID 4151974 The NBT test assesses the ability of immune cells to convert NBT to a blue dye. In chronic granulomatus disease, the immune cells are missing the ability to generate reactive oxygen via phagocyte NADPH oxidase, so those white blood cells fail the NBT test.
Performic acid PMID: 1099403
Cepko, C., Ryder, E., Fekete, D.M., and Bruhn, S. Detection of B-galactosidase and alkaline phosphatease activities in tissue. Online (archive): https://web.archive.org/web/20160821210733/http://genepath.med.harvard.edu/~cepko/protocol/xgalplap-stain.htm. An excellent brief discussion of X-Gal histochemistry. Much of the material presented here was derived using their references.
Studies in Enzyme Cytochemistry
I. Principles of cytochemical staining methods. Holt and Sullivan, Proc. R. Soc. London B 148 (1958) 465-480. A nice theortical discussion of reaction rates and diffusion rates through tissue as it relates to localization of a colormetric stain. Very useful as a tutorial on how to calculate diffusion, but not much help on LacZ staining per se. A key conclusion is that in order to get a good "localization factor" ( F ), the reaction rate generating the insoluble dye product must be much faster than the diffusion rate of the soluble substrate precursor. (therefore, oxidation rates of indoxyls must be increased with catalysts like ferricyanide).
II. Synthesis of indigogenic substrates for esterases. Holt and Sandler, Proc. R. Soc. London B 148 (1958) 481-494. Describes the synthesis and reactions of a series of indoxyls, including 5-bromo-4-chloro-indoxyl acetate (one of the indoxyl acetates(VII), Table 8 p. 493) . Note that acetate groups are attached to the indoxyls so that esterase enzymes could be used to rapidly form the dyes. Demonstrates that the NH group helps attach to local proteins to enhance intracellular localization.
III. Relationships between solubility, molecular association, and structure in Indigoid dyes. Holt and Sandler, Proc. R. Soc. London B 148 (1958) 495-505. the effects of different substitutions, configuation of the dimers, hydrogen bonding, molecular packing and crystallization on silutbility in water and different lipid environments. The presence of halogen substitutions cause less crystalline products (compared to indigo itself), which is good for a staining dye.
IV. Kinetics of aerial oxidation of indoxyl and some of its halogen derivatives. Cotson and Holt, Proc. R. Soc. London B 148 (1958) 506-519. Instead of an electron acceptor to aid in oxidation, oxygen itself can be used (aerial oxydation). Aerial oxidation requires a basic environment (i.e. pH 8.0 or above; p. 512). At pH 11.0, 87% of 5-bromo-4-chloro-indozyl should be converted to dye (one of the higher conversion rates reported; p. 514). Aerial oxidation also produces H202 as a by product, which might come in handy if an additional stain can be used to visualize the hydrogen peroxide (p. 515) . H202 causes a lot of artifacts, however, if there are peroxidases present, because the peroxidases will rapidly oxidase the indoxyls. There are two problems with aerial oxidation: 1. The lacZ enzyme prefers an acidic environment (pH 6.0), not a basic environment. 2. Aerial oxidation while fast is not fast enough to deposit the insoluble dye before it can diffuse (p. 518)
V. An appraisal of indigogenic reactions for esterase localization. Holt and Withers. Proc. R. Soc. London B 148 (1958) 520-532. This paper uses the best substrate identified in the preceding papers, 5-bromo-4-chloro-indoxyl acetate , to localize esterases in various rat tissues. The reactions were carried out at pH8.5 in Tris in 0.2 M NaCl using 5 mM potassum ferri- and ferrocyanides as oxidants (p. 522). At pH 6.0 and below, staining was diffuse even with high oxidant concntrations; basic conditions were best (pH 8.5 and above (p. 525-6). [Also tried aerial oxidation, with too much red blood cell staining and non specific staining; p. 524]. Notes that the NH group is important for protein binding, and the 5:4' substitutions enhance the binding to protein greatly (with disubstitutions, the dye leaves the cytoplasm and diffuses into fat.)
Note p. 523: "Exposure of stained sections to formalin vapour or solution must be very brief since formaldehyde reacts with many indigoid dyes to give soluble products and loss of stain."
Recommends ferrocyanide, and notes that it inhibits red blood cell staining "presumably because of the chemical similarity to cyanide"